Synthesis of 1,1'-Ferrocenedicarboxylic Acid
Apr 15, 2022
Introduction
Ferrocene is a ferric ion sandwiched between two upper and lower rings. Because of the aromaticity of the ring, the combination with ferric ions forms a stable sandwich structure. This stable structure is different from other organometallic compounds, mainly in that it is more stable, and the metal ions are not exposed and the ionicity is not strong. The ferrocene sandwiched ferrous iron is easily oxidized to ferric iron, which has strong electrochemical properties, and has many irreplaceable properties and bright application prospects in electrode modification and electrochemistry.
Ferrocene is relatively difficult to synthesize, and the cost of industrial production is high. However, the synthesis conditions of its derivatives are special and harsh, resulting in extremely high cost of industrial production and difficulty in mass production. It is even more difficult to improve the purity. The preparation of ferrocene derivatives is difficult and expensive.
Synthesis of CAS 1293-87-4 1,1'-FERROCENEDICARBOXYLIC ACID
1) Crude 1,1'-ferrocene dicarboxylic acid.
The target product 1,1'-ferrocene dicarboxylic acid is prepared by oxidizing 1,1'-diacetyl ferrocene. The branched acetyl group on ferrocene is easily oxidized, and the strength is above medium.
The oxidant can be used, but considering that the divalent iron ions in ferrocene are easily oxidized, the oxidant should not be too strong, and the separation of the product should be considered.
Finally, a NaClO solution with moderate oxidizing ability and easy product separation should be selected as the oxidant.
In the dark condition, a certain amount of 1,1'-diacetylferrocene was dissolved in NaClO solution with a mass fraction of 10%, reacted at 50 °C for 1 h, heated to 95 °C, and then stirred for 1 h. h. After cooling, a certain amount of NaClO solution with a mass fraction of 10% was added, and stirring was continued at a certain temperature, and the product solution was obtained after the reaction was completed. The solution of the product was filtered while hot, the filtrate was acidified with concentrated hydrochloric acid to pH 1~2, a large amount of yellow precipitate was formed, and the crude product was obtained after suction filtration.
2) Refined 1,1'-ferrocene dicarboxylic acid. The resulting product was placed in a beaker, and an appropriate amount of NaOH solution was added to dissolve the product as much as possible.
The solution was filtered, concentrated hydrochloric acid was added to the filtrate to make the pH value reach 1~2, and the filtrate appeared yellow precipitate. The filtrate was poured into a rotary evaporator and vacuumized for rotary evaporation. After evaporation to dryness, it was dissolved in absolute ethanol and evaporated naturally. After removing absolute ethanol, put it in an oven to dry to obtain refined 1,1'-ferrocene dicarboxylic acid.
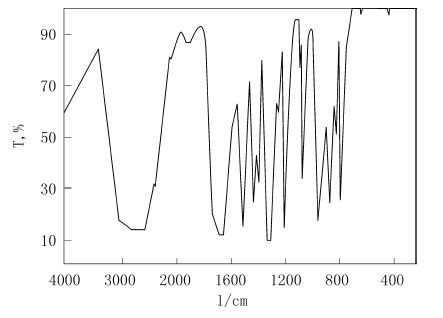
IR spectrum of 1,1'-ferrocenedicarboxylic acid
Conclusion
In this paper, the oxidation of 1,1'-diacetylferrocene to synthesize 1,1'-ferrocene dicarboxylic acid, the reaction temperature for the synthesis and the interaction between 1,1'-diacetylferrocene and sodium hypochlorite were studied. The molar ratio was investigated, and the optimal reaction temperature was 50 °C and the optimal molar ratio was 1:2.3. In this paper, the post-treatment was optimized to obtain 1,1'-ferrocene dicarboxylic acid with higher purity, and the product was confirmed as the target product by the determination of melting point and infrared.
You Might Also Like
-

CAS 82096-91-1 | 2-Chloro-4-Trifluoromethylbenzaldehyde
-

CAS 51114-68-2 | 3-CHLORO-O-ANISIDINE
-
![CAS 139306-10-8 | 3-[(1S)-1-(Dimethylaminoethyl)]phenol](/uploads/202235855/small/cas-139306-10-8-3-1s-1-dimethylaminoethyl48391456261.png?size=384x0)
CAS 139306-10-8 | 3-[(1S)-1-(Dimethylaminoethyl)]phenol
-

CAS 17878-44-3 | (4-BROMOPHENOXY)TRIMETHYLSILANE
-
CAS 3069-29-2 | 3-(2-Aminoethylamino)propyl-dimethoxymeth...
-

CAS 42292-18-2 | 3-Aminopropylbis(trimethylsiloxy)methyls...
